Deimmunization
Immune Engineering
Diminishing the immunogenicity of therapeutic proteins without hindering their function will improve clinical outcomes. This can be accomplished by substituting key amino acids within T cell epitope sequences which abrogate binding to HLA and thereby attenuate epitope potential to trigger a T cell response. Altered T cell epitopes no longer bind to HLA. Epitope modifications are easily evaluated in vitro and in vivo prior to release of the protein therapeutic for clinical development. EpiVax has proprietary technology to identify and introduce Tregitopes (T regulatory epitopes) to reduce immunogenicity and induce tolerance (patented technology/approach). This method of reducing immunogenicity, also known as de-immunization (DeFT), is described in further detail below.
DeFT: Deimmunization of Functional Therapeutics
The FDA has suggested that protein therapeutic developers assess and manage unwanted immunogenicity. DeFT™ is cost effective and highly accurate in establishing and eliminating the immunogenic risks hidden within your protein therapeutic or biologic. EpiVax’s deimmunization strategy is focused on the identification and elimination of T cell epitopes contained within your candidate sequence.
Established leaders, skilled team. EpiVax has developed and validated a streamlined approach to the deimmunization of therapeutic proteins, coupling advanced computational analysis with high throughput in vitro and in vivo assays. The team of scientists at EpiVax are recognized experts in immunogenicity, setting the pace in the deimmunization field with their unique mix of immunoinformatics and immunology.
Tools for deimmunization: the EpiMatrix system
Searching for Putative T cell Epitopes
EpiMatrix predicts the binding potential with respect to a panel of nine common Class II super-type alleles, covering the majority of the human population (>90%).
Searching for Epitope “Clusters”
We and others have found that potential immunogenicity is not randomly distributed throughout protein sequences but instead tends to “cluster” in immunogenic regions (which are often also immunodominant, see below), facilitating the process of deimmunization. The statistical algorithm ClustiMer allows us to screen EpiMatrix results sets and identify putative T cell epitope clusters.
Evaluating Overall Immunogenic Potential
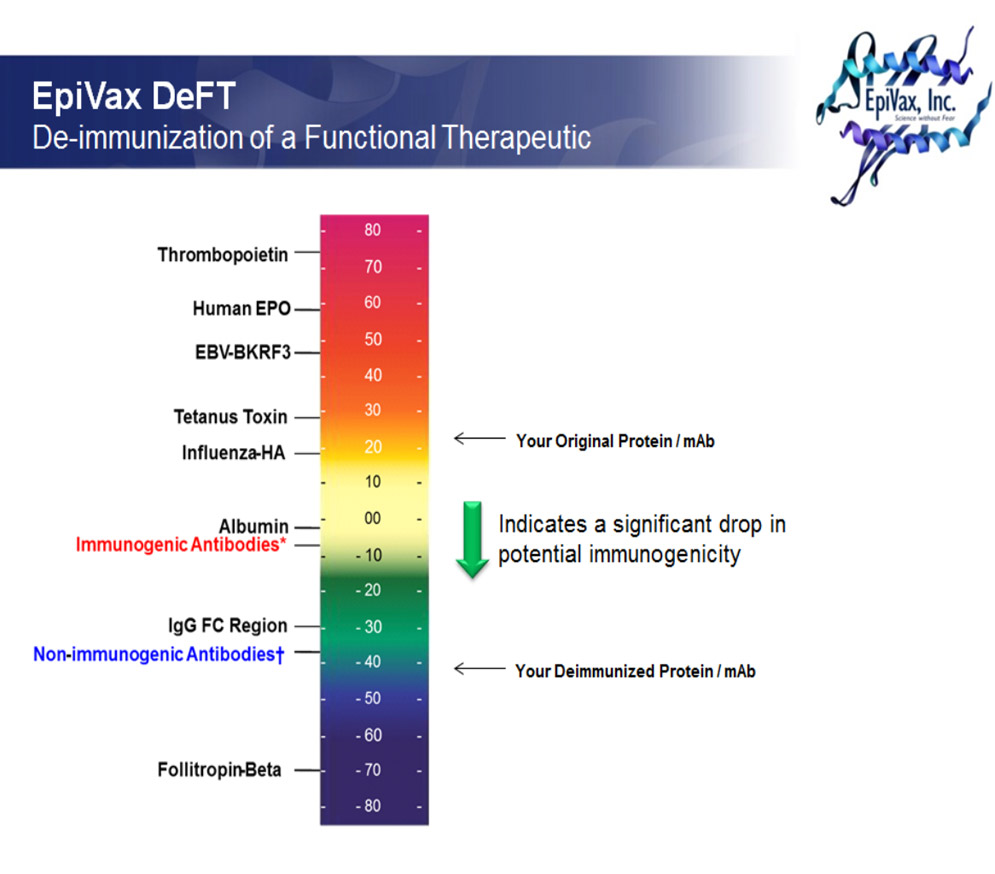
The ability to compare peptide scores across multiple alleles, a feature unique to the EpiMatrix system, is the key to evaluating the overall immunogenic potential of proteins. For a given protein, the frequency and number of observed hits compared to expected hits is a good indicator of overall immunogenic potential. Our expectation is based on an EpiMatrix analysis of a large sample of randomly generated sequences with normal amino acid frequencies; the expectation is expressed as hits per 1000 amino acids to account for the variable sizes of target proteins. Examples of autologous proteins that score low on the scale include albumin and insulin.
Evaluating Regional Immunogenic Potential
Within proteins, regional immunogenic potential (T cell epitope clusters) can be quickly assessed. We have also noted a characteristic pattern that we call the “EpiBar” (Figure 1). This band-like pattern, in which a single 9-mer frame scores well among four or more ‘super-type’ alleles, is characteristic of promiscuous epitopes. Immunodominant sequences that contain EpiBars include Tetanus Toxin 825-850 (deviation +16), Flu HA 306-318 (deviation +18) and GAD65 557-567 (deviation +19). When selecting regions for deimmunization, we give preference to immunodominant epitopes that also contain this EpiBar pattern.

Figure 1: EpiMatrix Cluster analysis. As an example, consider the flu HA peptide, an epitope known to be promiscuously immunogenic. It scores extremely high for all nine alleles in EpiMatrix. The deviation from a value expected for a random sequence is + 17.62. Deviation scores greater than 10 are significant and considered to be likely promiscuous epitopes. The high scores across alleles in the third frame of this peptide form a pattern known as an EpiBar. This band-like pattern is characteristic of promiscuous epitopes.
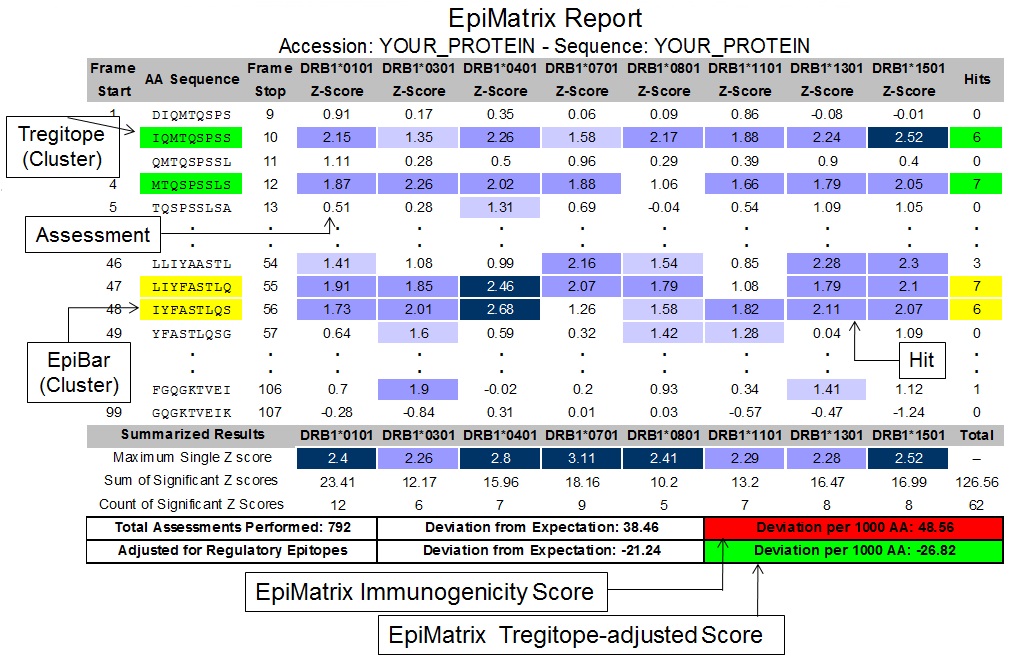
Figure 2: Identifying effector epitope clusters (regional immunogenic potential in yellow) within whole proteins
Potential immunogenicity is not evenly distributed across the length of a protein but tends to cluster. Each overlapping 9-mer frame (rows) is given a score for likelihood to bind with each of nine common Class II HLA alleles (columns). Medium blue shading indicates a score in the top 5% and dark blue shading indicates a score in the top 1%. Blue shading indicates all other scores (neutral); scores in the top 10% (but below the top 5%) are shown but are not considered significant. Blue outline indicates the span of the cluster. EpiVax also (uniquely) identifies Tregitopes (T regulatory epitopes) in green that may reduce immunogenicity)

Figure 3: OptiMatrix “logo” report showing individual contribution to binding averaged across alleles and frames. The size and color of the amino acid residue is keyed to its contribution to binding across frames for nine selected class II alleles (see below). OptiMatrix scores are also shown.
Designing Deimmunized Sequences
To identify amino acids within clusters that are suitable for modification, we first create a “logo report” (Figure 3) for each of the immunogenic regions using OptiMatrix. These reports identify “critical” residues, which contribute most to MHC binding.
OptiMatrix offers the mutation with the greatest impact. Also, one can independently choose mutations. In making our selections, we generally consider common mutations, changes tolerated in other species or variants of the target protein, published information describing residues important to structure and/or function and, if structural models are available, predicted impacts on tertiary structure. We then focus on “key” amino acids, illustrated by a larger font size (and higher OptiMatrix score), in the logo report. Given this list of targeted amino acids and viable alternatives we then evaluate all possible alternative sequences and compile a list of the best single amino acid changes, the best double changes, the best triple changes, and, if necessary, even more complex changes, using OptiMatrix. The de-immunized sequences are then validated (in vitro) before being integrated into the protein and tested for functionality (in-vivo).
Limiting Modifications to Immunodominant Regions
The connection between immunodominance and promiscuous binding has been illustrated in several proteins. EpiVax focuses on modifying immunodominant determinants, not on modifying all peptides capable of binding to class II HLA. In contrast to methods others have pursued, our proprietary technology allows us to limit the modifications to one or two amino acids per immunodominant region thus further reducing the impact on protein function. EpiVax has demonstrated that modification of a few immunodominant epitopes with promiscuous binding reduces immunogenicity of protein therapeutics (this is known as the “threshold effect”). This has also been demonstrated in a study of erythropoietin: reduced immunogenicity was observed when modifications were made to only two regions of the protein. In that case the sequence changes reduced both HLA binding affinity and human T cell responses while having no significant effect on bioactivity.
Target Validation and Deimmunization
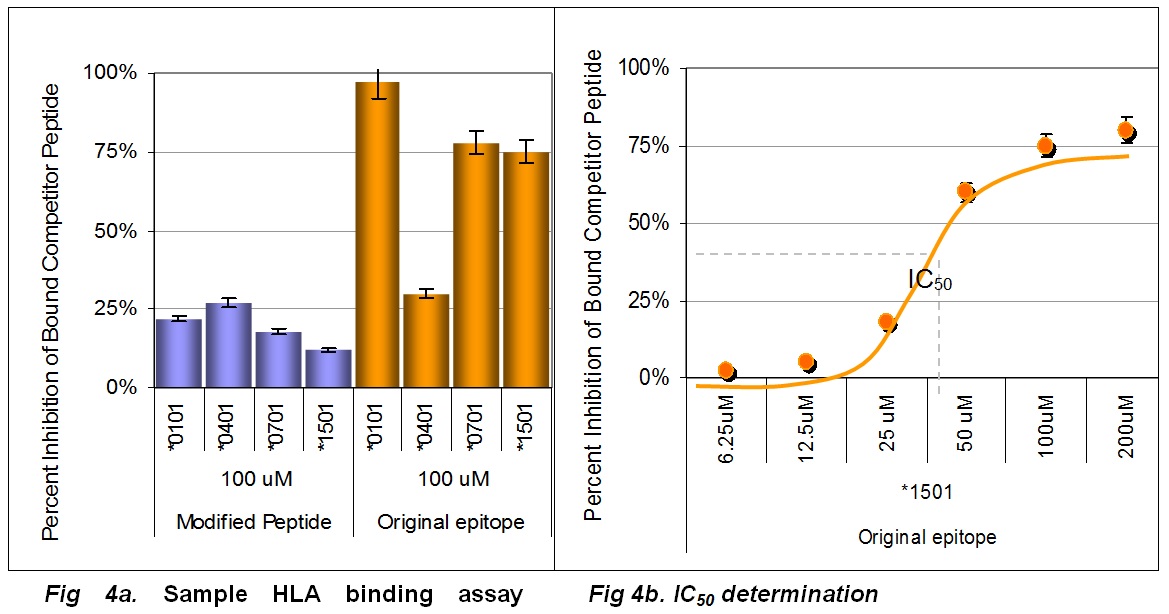
Each of the identified epitopes and corresponding variants are tested as peptides in-vitro for binding affinity with respect to a panel of MHC Class II alleles. The HLA cell free binding assay serves as a “first pass” screen for confirming epitopes and de-immunized peptides. Any cluster that is not confirmed to bind is not in need of deimmunizing modifications.
In a binding assay screen, an unmodified peptide and its modified counterparts are compared in their binding affinity for five HLA alleles. The unmodified (gold bars) peptide binds with high affinity across three of four tested HLA alleles. As intended, strategic modifications to this sequence confer disruption of binding (blue bars). Failure to bind to HLA is usually indicative of lower immunogenicity in T cell assays.
Modification, Re-synthesis, Expression. The next step in the deimmunization process is to integrate the modifications present in the de-immunized T cell epitope clusters into the full-length protein. Expressed proteins are purified, tested for function in vitro and for immunogenicity in HLA transgenic mice. Over the past years, we have been engaged in a number of protein re-engineering projects using the DeFT approach. Information about our successful DeFT programs will be provided upon request.
Tregitopes, a proprietary tool for DeFT:
Tregitopes are a set of peptides that EpiVax discovered using the EpiMatrix platform while screening large libraries of IgG antibodies. They have the ability to call off the body’s immune response and can induce tolerance to co-administered antigens. When appropriate, Tregitopes can be incorporated into the DeFT process. Tregitopes are a set of peptides that specifically activate CD4+CD25+FoxP3+ natural regulatory T cells (nTregs). Tregitopes are promiscuous MHC Class II T cell epitopes located in the Fc and framework regions of Fab from IgG. In vitro, co-incubation of antigens with Tregitopes leads to suppression of effector cytokine and chemokine secretion, reduced proliferation of effector T cells, and expansion of antigen-specific adaptive Tregs (aTregs). In vivo, co-administration of Tregitopes with a wide range of proteins (such as FVIII, thyroid stimulating hormone receptor, ovalbumin, and autoantigens) leads to suppression of T cell and antibody responses to test antigens. Review our full list of Tregitope publications with links to full PDFs.
We are dedicated to applying our tools (in-silico, in-vitro and in-vivo) to re-engineering therapeutic proteins and to designing new vaccines. For a recent interview highlighting our CEO’s latest ideas, click on the video:
![]()